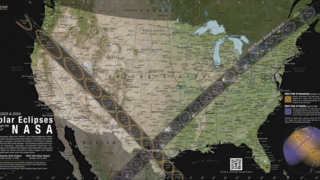
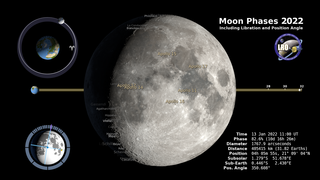
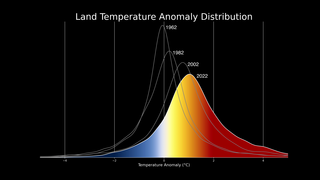
Earth
ID: 825
Ozone is very reactive. It easily loses the third oxygen atom in the presence of other highly reactive compounds called radicals, which contain chlorine, hydrogen, nitrogen, or bromine. Minute quantities of these radicals can cause large decreases in ozone because they are not consumed in the reaction. This is called a catalytic cycle.


Chemical Model Animation of O3 Losing an Oxygen Atom to a Radical


For More Information
Visualization Credits
James W. Williams (GST): Lead Animator
Jesse Allen (Raytheon): Animator
Greg Shirah (NASA/GSFC): Animator
Mark Schoeberl (NASA/GSFC): Scientist
Jesse Allen (Raytheon): Animator
Greg Shirah (NASA/GSFC): Animator
Mark Schoeberl (NASA/GSFC): Scientist
Please give credit for this item to:
NASA/Goddard Space Flight Center Scientific Visualization Studio
NASA/Goddard Space Flight Center Scientific Visualization Studio
Short URL to share this page:
https://svs.gsfc.nasa.gov/825
This item is part of this series:
UARS
Keywords:
SVS >> Oxygen
GCMD >> Earth Science >> Atmosphere >> Atmospheric Chemistry/Oxygen Compounds >> Ozone
NASA Science >> Earth
GCMD keywords can be found on the Internet with the following citation: Olsen, L.M., G. Major, K. Shein, J. Scialdone, S. Ritz, T. Stevens, M. Morahan, A. Aleman, R. Vogel, S. Leicester, H. Weir, M. Meaux, S. Grebas, C.Solomon, M. Holland, T. Northcutt, R. A. Restrepo, R. Bilodeau, 2013. NASA/Global Change Master Directory (GCMD) Earth Science Keywords. Version 8.0.0.0.0
https://svs.gsfc.nasa.gov/825
This item is part of this series:
UARS
Keywords:
SVS >> Oxygen
GCMD >> Earth Science >> Atmosphere >> Atmospheric Chemistry/Oxygen Compounds >> Ozone
NASA Science >> Earth
GCMD keywords can be found on the Internet with the following citation: Olsen, L.M., G. Major, K. Shein, J. Scialdone, S. Ritz, T. Stevens, M. Morahan, A. Aleman, R. Vogel, S. Leicester, H. Weir, M. Meaux, S. Grebas, C.Solomon, M. Holland, T. Northcutt, R. A. Restrepo, R. Bilodeau, 2013. NASA/Global Change Master Directory (GCMD) Earth Science Keywords. Version 8.0.0.0.0